Phosphorus Electron Configuration | A Simple Guide On Writing and Understanding It
The electron configuration of important Phosphorus as an element is one of the basic building blocks while learning chemistry and is valuable in understanding the phosphorus nonmetals useful in farming and Chemistry subject fields which give an insight of its placement in group 15 of the periodic table. If you are studying, being able to write the electron configuration for phosphorus and comparing it with other elements is bound to help.
In this blog, we will talk about how to write phosphorus’s electron configuration, review some related common configurations, and learn about phosphorus’s position in the periodic table.
How to write general configuration of phosphorus?
The steps to take when writing the electron configuration phosphorus are straightforward if you understand these facts:
- The atomic number of phosphorus is 15.
- So a neutral phosphorus atom is 15 electrons.
- According to the Aufbau Principle, electrons fill orbitals starting from the lowest energy level upwards.
In a Sequential Manner:
- Beginning with the 1s orbital → 2 electrons
- Next 2s orbital → 2 electrons
- Next, 2p orbital → 6 electrons
- Next, 3s orbital → 2 electrons
- Next, fill 3p orbital → 3 electrons
Final Electron Configuration for Phorus
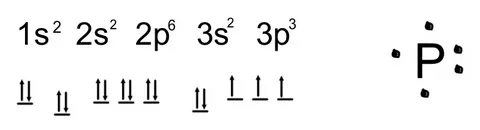
1s² 2s² 2p⁶ 3s² 3p³
And so it has five valence electrons (3s² 3p³) which explains much of its chemical behavior including forming three covalent bonds in many compounds.
Understanding phosphorus’ position on the periodic table
Phosphorus belongs to:
- Group 15 (also known as the nitrogen group)
- Period 3
- Atomic number: 15
- Symbol: P
Very versatile is phosphorus as working in biology (in DNA, ATP and phospholipids) and in industry as (fertilizers and phosphorus in detergents).
Which element has the electron configuration of 1s² 2s² 2p⁶ 3s² 3p²?
Let’s see step by step.
- 1s² → 2 electrons
- 2s² → 2 electrons
- 2p⁶ → 6 electrons
- 3s² → 2 electrons
- 3p² → 2 electrons
Total = 2 + 2 + 6 + 2 + 2 = 14
An atom with this many electrons is Silicon (Si).
Answer:
Si has the electron configuration as such 1s² 2s² 2p⁶ 3s² 3p².
What is the element corresponding to the electron configuration 1s² 2s² 2p⁶ 3s² 3p⁵?
Let’s simplify:
- 1s² = 2
- 2s² = 2
- 2p⁶ = 6
- 3s² = 2
- 3p⁵ = 5
Adding the total: 2 + 2 + 6 + 2 + 5, equals 17 electrons.
This means the element with 17 electrons must be Chlorine (Cl).
Conclusion:
As calculated, Chlorine (Cl) has the electron configuration 1s² 2s² 2p⁶ 3s² 3p⁵.
Chlorine has seven valence electrons that are easily lost or shared with other elements during chemical reactions, resulting in the formation of compounds, for example NaCl (table salt), therefore making it highly reactive.
Which element has the following electron configuration: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d⁵?
Let’s break this down:
- 1s² = 2
- 2s² = 2
- 2p⁶ = 6
- 3s² = 2
- 3p⁶ = 6
- 4s² = 2
3d⁵ = 5
2 + 2 + 6 + 2 + 6 + 2 + 5 equals 25 electrons, and means the atom with 25 electrons is Manganese (Mn).
Answer:
Manganese (Mn) has the electron configuration: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d⁵.
Manganese is a transition element and, owing to the incompletely filled d subshell, exhibits several oxidation states.
Noble gas shorthand for phosphorus
- We can adopt noble gas shorthand to ease the simplification of phosphorus’ electron configuration.
The shorthand of electron configuration for phosphorus would be:
The noble gas preceding phosphorus is Neon (Ne) → 1s² 2s² 2p⁶.
[Ne] 3s² 3p³
This format is particularly beneficial for larger atoms needing simplification due to extensive electron configuration sequences.
The impact of electron configuration on the chemistry of phosphorus
Phosphorus can easily participate in chemical reactions due to the five valence electrons (3s² 3p³) where it can:
- Form three covalent bonds (e.g., in phosphine PH₃)
- Exhibit multiple oxidation states: -3, +3, +5
- Participate in double bonds with oxygen in compounds such as phosphoric acid (H₃PO₄)
- Gaining three electrons for ionic bonds (e.g., forming P³⁻ ion)
Phosphorus compounds, along with starving organisms, are crucial due to their essential roles for energy stroke and transfer (ATP) and the structural framework of DNA.
Comparison of Phosphorus with Silicon, Manganese and Chlorine
| Property | Phosphorus (P) | Silicon (Si) | Chlorine (Cl) | Manganese (Mn) |
| Atomic Number | 15 | 14 | 17 | 25 |
| Group | 15 | 14 | 17 | 7 (transition) |
| Valence Electrons | 5 | 4 | 7 | Varies (d-orbital) |
| Typical Bonds | 3 covalent bonds | 4 covalent bonds | 1 covalent/ionic bond | Multiple oxidation states |
| Role | DNA, ATP, fertilisers | Semiconductors | Salts, disinfectants | Steel production, catalysis |
Understanding electron configurations of each element explains this elemental behavior despite their proximity on the table.
Mistakes when focusing on phosphorus
- Order of filling is wrong: do not forget 3s fills before 3p.
- Forgetting to switch orbitals: 2p comes before 3s.
- Octet mistakes: Aiming for 8 in some reactions.
- Config. shorthand: Phosphorus would be [Ne].
Explain configuration importance:
Predicted patterns include:
- Bonding
- Reactivity
- Config of matter (solid, liquid, gas)
- Electrical conduction
- Magnetic conductivity.
Explaining phosphorus’ electron configuration in simple terms.
The valency of phosphorus is indicated as 1s² 2s² 2p⁶ 3s² 3p³, where it has five outer shell electrons waiting to bond. Understanding the reason phosphorus acts a certain way, either in organisms or in materials that surround us, differs from region to region and requires knowing why configurations are written as such.
Identifying the element of configurations such as 1s² 2s² 2p⁶ 3s² 3p², 1s² 2s² 2p⁶ 3s² 3p⁵, and 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d⁵ strengthens your understanding of periodic trends alongside the chemical behavior as a whole.
Phosphorus provides an incredible starting point to grasping the energizing world of chemistry and mastering electrons’ configurations.




Post Comment