Exploring the Trends of Electron Affinity Across the Periodic Table
One of the most interesting phenomena to jump into when considering the periodic table is electron affinity. It’s not merely about the acquisition of an electron; it is a whole saga about the elements, how they are intertwined, and how they partake in reactions and bonds.
In this tutorial, we will discuss the changing trends of electron affinity as one moves across the periodic table, the reasons behind such features, and how these concepts differ in meaning to electronegativity. This explanation is bound to assist a learner or an inquisitive individual familiar at some level with chemistry to grasp the logic behind the behavior of electrons.
Definition of Electron Affinity
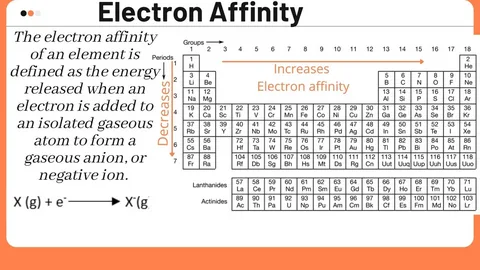
The term “affinity” refers to the energy change that occurs when an atom in a gaseous state captures an electron. If some energy is released during the process, then the affinity is said to be negative (which means energy is exothermic). The greater energy released, the greater will be an atom’s desire towards to possess that extra electron.
Simply put, an atom’s eagerness in accepting an electron is defined as the atom’s electron affinity.
Does electron affinity increase from left to right?
Yes, it does increase, in an overall manner. But why?
The arguments that were just made, plus the following points:
- The number of protons in the nucleus increases (positive charge).
- The atomic radius gets smaller (positive charge).
- The attraction between the nucleus and the added electrons is stronger (positive charge).
The aforementioned make the right side of the periodic table (in particular the halogens) far more likely to accept an electron in comparison to the left-hand side, which contains the alkali metals.
So, does “electron affinity increase from left to right?” can be answered as yes with some exceptions like noble gases and certain transition metals.
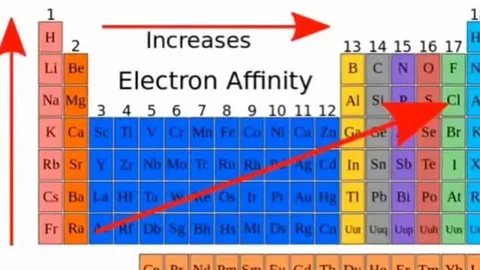
Why would electron affinity increase down a group?
Actually, this is a trick question.
It is widely accepted that the notion “affinity of electrons increases down a group” is wrong for most scenarios. In reality, it is much more common that electron affinity decreases as you go down a group.
Why?
Increased atomic size: The electrons are added at increasing distances from the nucleus.
Increased shielding: Inner electrons block nuclear attraction to the incoming electron.
Reduced nuclear attraction: The atom is less able to attract and hold onto an extra electron.
In the same group (Group 17), iodine and fluorine are examples of this because fluorine has a higher electron affinity. Thus, the correct trend is: electron affinity becomes less favorable as you move down a group.
This explains the statement “Why does electron affinity increase down a group?”— it does not! More often than not, it declines.
What is the difference between electron affinity and electronegativity?
Let’s resolve this mix-up— what is the difference between electron affinity and electronegativity?
There are distinctions, even if they appear related:
| Property | Electron Affinity | Electronegativity |
| Definition | Energy released when an atom gains an electron | Ability of an atom to attract shared electrons |
| Measured in | kJ/mol (energy units) | No units (relative scale) |
| Involves | Isolated atoms | Atoms in a chemical bond |
| Affected by | Nuclear charge, size, electron configuration | Same factors + bonding behavior |
Who has the highest electron affinity?
Addressing an interesting problem: Who has the highest electron affinity?
Chlorine holds the record for the most negative electron affinity, so chlorine wins this one.
Chlorine has a slightly higher electron affinity than fluorine–which seems odd considering that chlorine is less electronegative than fluorine–for the reasons below:
- The repulsion from adding an extra electron is much too strong for fluorine’s diminutive outer shell.
- Chlorine is a little larger than fluorine allowing for some extra room when adding an incoming electron, making energy release more favorable.
If someone asks just like you, who has the lowest electron affinity now, the answer is readily available–it’s Chlorine.
Does electron affinity increase from up to down?
This is a common misunderstanding.
Moving down a group in the periodic table, the value for electron affinity decreases is the correct tendency. So no is an appropriate answer for “Does electron affinity increase from up to down?”
Top group elements such as your oxygen and fluorine will find gaining an additional electron much easier than the lower group elements like sulfur and iodine.
Why do electrons appear to be moving from a higher potential energy zone to a lower one (left to right)?
In chemistry, “why does it appear that electrons flow from left to right?” is a more encompassing question which is integrated into electric circuits. However, when looking at it from the view of periodic trends:
- Metals like sodium and potassium are situated to the left of the periodic table, which means they easily lose electrons as they are metals.
- To the right, nonmetals like Chlorine and fluorine are nonmetals as they gain electrons.
So, there is a metaphorical ‘flow’ of electrons from metals (left) to nonmetals (right) especially in ionic bonding.
It’s all about balance: nature seeks to equalize ‘charges’ where metals give it out and nonmetals accept it.
What is the trend in electron affinity between groups?
Looking at group differences, let’s investigate what is the trend in electron affinity between groups:
Alkali metals (group 1) are known to have very low electron affinities. Losing electrons is much more favorable for them, so 1 as a result is the preferred number.
- From group 17 (halogens), elements have very high electron affinities, as they are overly eager to get one more electron to complete their octet.
- Almost no electron affinity is found from group 18 (noble gases) since they already have full outer shells.
- It’s a set in stone rule, moving left to right across any period, electron affinity should increase, but within different groups elements from group 17 are the winners whereas group 1 and 18 are the losers.
Does electronegativity increase from left to right?
Yes! Identical to the trend of electron affinity, electronegativity increases from left to right across a period.
Why?
- The number of protons increases
- Atomic radius decreases
- Attraction between the nucleus and bonding electrons strengthens
This trend is also the reason why elements such as fluorine and oxygen are significantly electronegative; they are small in size and have considerable nuclear attraction.
So in response to the keyword “Does electronegativity increase from left to right?” Yes, absolutely.
Does electron affinity increase or decrease down Group 7?
Group 7 is also the halogen group which includes:
- Fluorine
- Chlorine
- Bromine
- Iodine
- Astatine
These elements have been characterized by having a high electron affinity but does electron affinity increase or decrease down Group 7?
It decreases.
As you move down the group 7:
- Atoms become larger.
- Electrons on the outside are further away from the nucleus.
- The ability to attract a new electron becomes significantly weaker.
Hence why fluorine and chlorine can attract electrons more readily than bromine or iodine.
Summary of trends of electron affinity
So, here is a summary of the most crucial things you have learned:
- Electron affinity receives maximum value in group 17 and minimum in group 1.
- Electron affinity increases from left to right across periods.
- Electron affinity decreases from top to bottom in a group.
- Elements in group 17 (halogen) have the highest electron affinities.
- Chlorine is known to have the greatest electron affinity out of all elements.
- While differing in definition, electron affinity and electronegativity are closely linked.
- In general, non-metals tend to gain electrons while metals lose them.
- Real-life relevance of electron affinity
Real-life relevance of electron affinity
All of this raises the question: “Why does this matter?”
The following are ways in which electron affinity is important:
- Determining chemical bonds and predicting chemical activity Zealous components are often subjected to gaining electrons. This makes identifying areas of reactivity much simpler.
- Bonds through understanding focus – a marked tendency to gain negative ions also permits the formation of anions.
- Crafting materials semiconductors require carefully tailored electronic functions to perform. These components are often used in batteries due to needing selected elements.
- Explaining processes Some of the trace elements in the organism behave according to these hypotheses.
Despite being a concept tackled in chemistry lessons, electron affinity determines the elements and processes upheld in the modern world.
Final thoughts
The period table functions as more than just a chart. It serves as a collection of elements and their interactions, and one of the main principles of atomic interactions is electron affinity.
In our strive to ease the use of a periodic table, we have answered, “Does electron affinity increase from left to right?” and “What is electron affinity vs electronegativity?” both fundamental questions to electron affinity.
Knowing trends like these provides insights into the microscopic world that shapes the materials we touch, the medicines we consume, and the technology we utilize every day.



Post Comment