Clearing the Confusion | Nitrogen Valence Electrons – 3 or 5?
Nitrogen is one of the most important elements in life and chemistry. It is present in DNA and proteins like air, which means it is more than just another element. One aspect of its chemistry tends to confuse a lot of individuals, though.
“Does nitrogen have 3 or 5 valence electrons?”
In this article, we aim to disclose the mystery of dual valency while explaining everything you need to know regarding nitrogen’s family and its periodic table’s constituents.
How many electrons does nitrogen have?
Nitrogen’s valence electrons are better assessed through the basic building blocks of chemistry, namely electrons.
Does nitrogen retain 7 electrons?
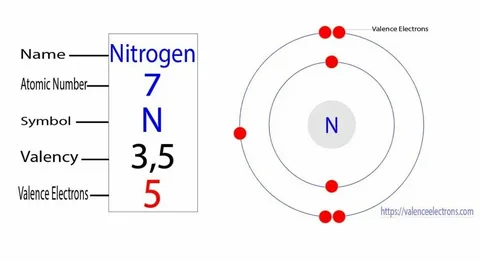
Yes. With an atomic number of 7, nitrogen’s neutral atom incorporates 7 electrons – which are 2 in the first shell and 5 in the second.
Electron configuration:
1s2 2s2 2p3
This establishes 5 electrons to be present in the outermost or 2nd shell, and these are valence electrons.
Does nitrogen have 3 or 5 valence electrons?
“Does nitrogen have 3 or 5 valence electrons?”
Direct answer is that nitrogen possesses 5 valence electrons.
The confusion sets in because the normal valency of nitrogen is 3.
Valence electrons = Are the electrons located at the outermost shell of an atom
Valency = the electrons which an atom is ready to give, share or accept to achieve a stable state
Hence nitrogen has 5 valence electrons but usually shares 3 of them during chemical bonding hence its typical valency is 3.
Is the Valency of Nitrogen 3 or 3?
This looks like a typing error (might be a case of asking if Nitrogen’s valency is 3 or 5) but let’s tackle it that way.
Is the valency of nitrogen 3 or 3?
It’s 3 for most folks with no debate in section compounds.
In the least branched structure, nitrogen has to gain 3 electrons to fill its octet (8 electrons in the outer shell). It usually makes 3 covalent bonds like in ammonia (NH₃) in which nitrogen shares three of his five valances.
Why Is Nitrogen Valency 5?
Now here’s a turn
Why Is Nitrogen Valency 5?
For some compounds, like nitric acid (HNO₃) or the nitronium ion (NO₂⁺), nitrogen can exhibit a valence of 5 because it uses d-orbitals to expand the octet. Heavier elements are easier to explain this for.
As an example sufficing for the coordinate or resonance bonding paradigms, for nitrogen, a second-period element without accessible d-orbitals, the valency of 5 is found in certain compounds. These are exceptions, not the norm.
Hence, while for practical reasoning, we may say nitrogen possesses a valency of 5, in reality, it has a standard valency of 3.
Which Element Has 5 Valence Electrons?
Beside nitrogen, other elements also possess 5 valence electrons. Nitrogen lays in Group 15 (or Group V) of the periodic table—famously known as the nitrogen family or the pnictogens.
So, “Which element has 5 valence electrons?”
The mentioned constituents of this group are the following:
- Nitrogen (N)
- Phosphorus (P)
- Arsenic (As)
- Antimony (Sb)
- Bismuth (Bi)
All above mentioned have 5 electrons in the outermost electron shell, thus they usually exhibit analogous bonding behavior, though there are anomalies such as the metals Sb and Bi which also show other valencies.Which Elements Have 5 Valence Electrons in the Nitrogen Family?
To narrow down, here is the answer to the specific focus keyword:
Which elements have 5 valence electrons in the nitrogen family?
These five:
| Element | Symbol | Atomic Number |
| Nitrogen | N | 7 |
| Phosphorus | P | 15 |
| Arsenic | As | 33 |
| Antimony | Sb | 51 |
| Bismuth | Bi | 83 |
They all have 5 valence electrons but their physical and chemical properties are different due to their position in the group and their metallic or nonmetal nature.
What Has 7 Valence Electrons?
You might wonder:
“What has 7 valence electrons?”
That would be the elements from group 17, the halogens. They are:
- Fluorine (F)
- Chlorine (Cl)
- Bromine (Br)
- Iodine (I)
- Astatine (At)
They all have seven electrons in the outer shell and usually need one more electron for stability.
On the other hand, nitrogen with 5 valence electrons will be gaining three or sharing three electrons to become stable.
Who Has A Valency Of 3 And 5?
Let me answer this question for you as it appears someone has drawn your interest;
Whose valency is 3 and 5?
There is nitrogen, phosphorus, arsenic, antimony and bismuth. Major 15 group elements of the periodic table exhibit both 3 and 5 valencies.
Take nitrogen for instance: it has NF3 where it has a valency of 3 and N2O5 where it has a valency of 5.
Moving on ditto of phosphorus there exists PCl3 and PCl5.
Because big atoms have various bonding situations and available orbitals, the variability occurs. With nitrogen though, however, less common and complex bonds are needed to populate 5, so it’s rare to value 5.
What Is The Lightest Element?
Now, before I forget, you posed the questions;
What is the lightest element?
As far as I know the lightest element is hydrogen (H).
- Atomic number: 1
- Atomic mass: 1.008 u
- Valence electrons: 1
Like other light elements, atomic hydrogen serves mainly as a reductant in acids due to being highly reactive, and valency 1 is seldom if not ever used. It does however play an important role in molecular unit formation and serves as a way simpler example when teaching about chemical bonds.
Facts Regarding the Valency of Nitrogen
Let us summarize the facts concerning the valence of nitrogen and its chemistry:
| Question | Answer |
| Does nitrogen have 3 or 5 valence electrons? | It has 5 valence electrons |
| Is the valency of nitrogen 3 or 3? | Likely meant 3 or 5 – it’s 3 |
| Why is nitrogen valency 5? | Rare; occurs in special bonding cases |
| Does nitrogen always have 7 electrons? | Yes, in a neutral atom |
| What has 5 valence electrons? | Nitrogen family elements |
| Whose valency is 3 and 5? | Group 15 elements like P, As, Sb |
| What has 7 valence electrons? | Halogens (F, Cl, Br, etc.) |
| What is the lightest element? | Hydrogen (H) |
| Which elements have 5 valence electrons in nitrogen group? | N, P, As, Sb, Bi |
Final Thoughts
So, now if someone asks, “Is nitrogen valency 3 or 5?”, we can confidently claim both, but primarily 3 for the majority of compounds. Nitrogen does have 5 valence electrons, but does not always use all of them when bonding.
These are some of the reasons why learning the behavior of nitrogen in different chemical contexts is not solely a class lesson in chemistry, it serves as a basis in learning biology, environmental science, and even materials engineering.
Nitrogen’s range of uses, from forming the backbone of proteins to stabilising explosives, is astonishing. Knowing how and when it uses its electrons allows one to understand simple substances such as NH₃ and more complicated ones such as HNO₃.


Post Comment